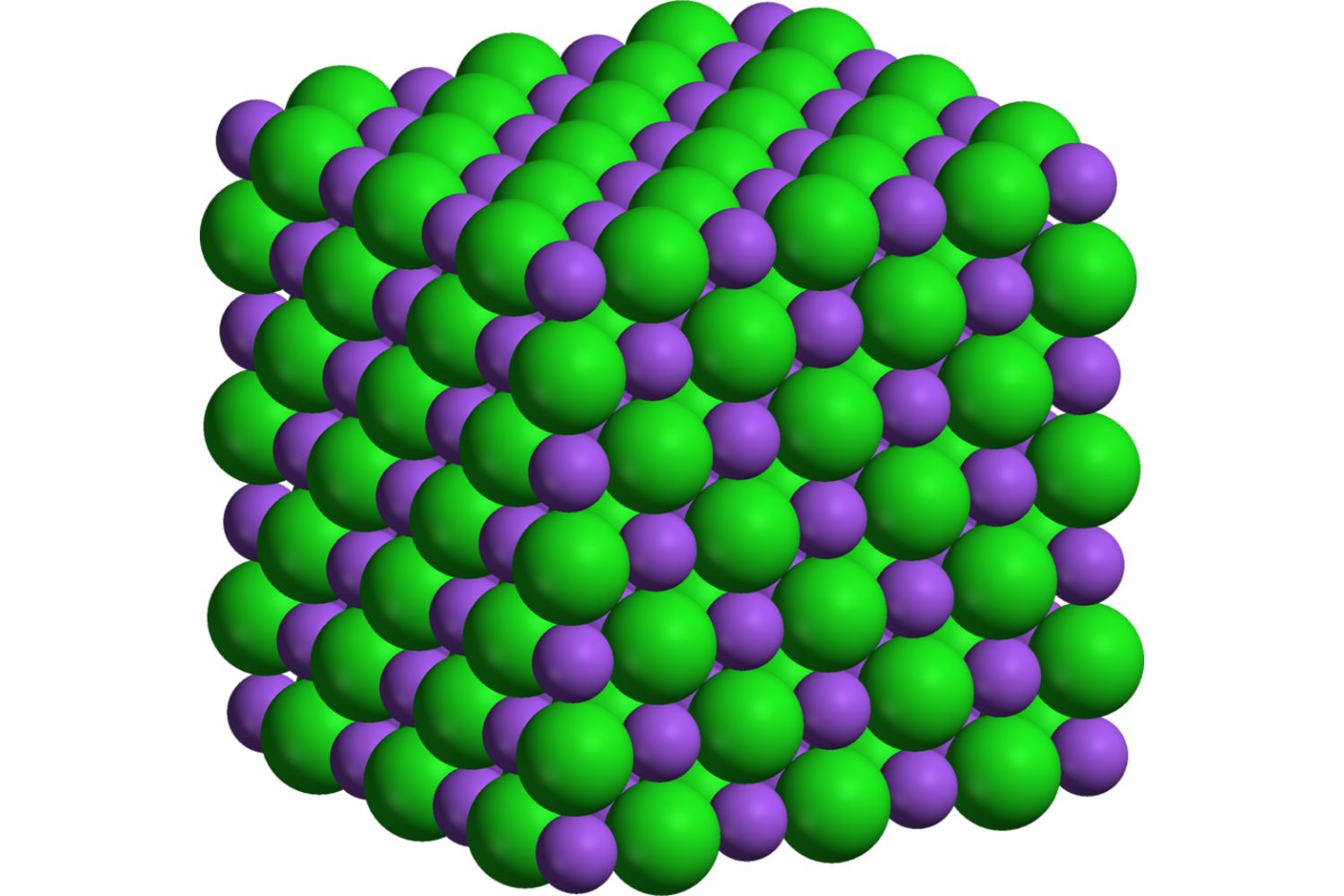
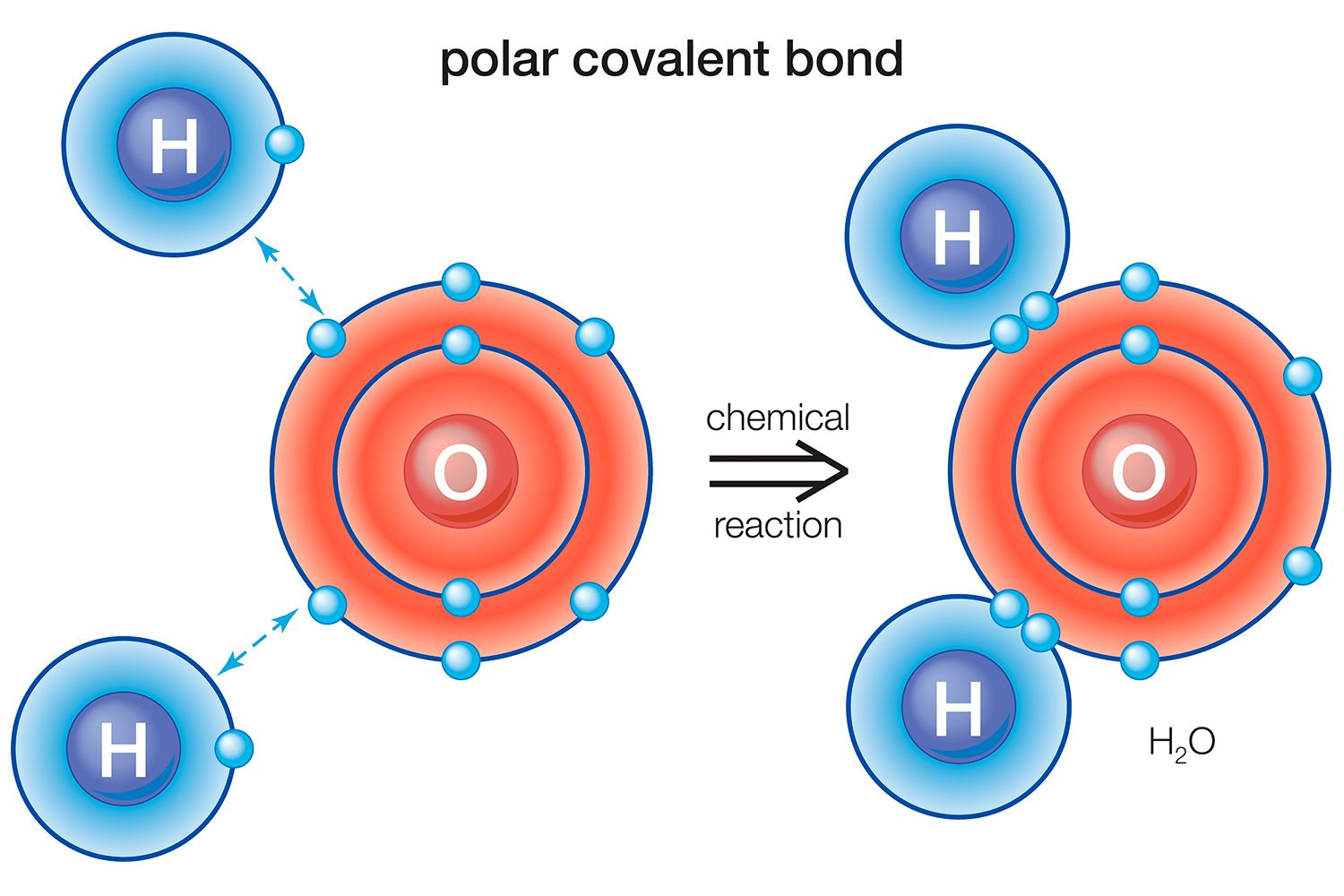
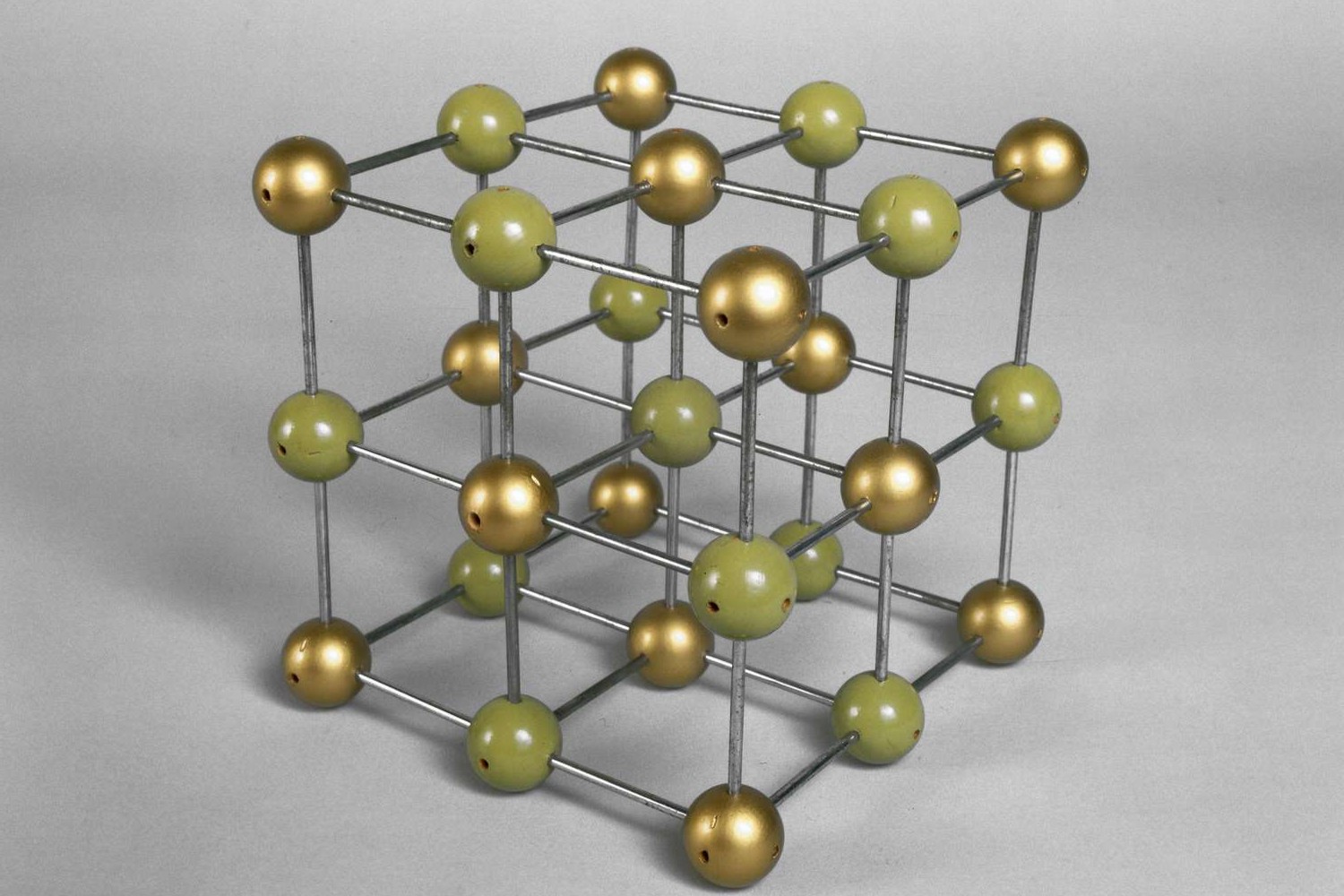
Dive into the world of ionic bonding, where atoms exchange electrons to create stable compounds. This quiz will test your grasp on the principles and intricacies of ionic bonds, from their formation to their properties. Ready to prove your chemistry prowess? Let’s get started!
We recommend that you do not leave the page that you are taking this quiz in. Stay honest 🙂
Ionic Bonding Quiz Questions Overview
1. What is an ionic bond?
A bond formed by the sharing of electrons between atoms
A bond formed by the transfer of electrons from one atom to another
A bond formed by the sharing of protons between atoms
A bond formed by the transfer of protons from one atom to another
2. Which of the following pairs of elements is most likely to form an ionic bond?
Sodium and Chlorine
Hydrogen and Oxygen
Carbon and Hydrogen
Nitrogen and Oxygen
3. What is the charge on the sodium ion in sodium chloride (NaCl)?
0
+1
-1
+2
4. What is the charge on the chloride ion in sodium chloride (NaCl)?
0
+1
-1
+2
5. Which property is commonly associated with ionic compounds?
Low melting points
Good electrical conductivity in solid state
High melting points
Poor solubility in water
6. In an ionic bond, which type of element typically loses electrons?
Non-metals
Metals
Noble gases
Metalloids
7. In an ionic bond, which type of element typically gains electrons?
Non-metals
Metals
Noble gases
Metalloids
8. Which of the following is an example of an ionic compound?
H2O
CO2
NaCl
CH4
9. What happens to the electrons in an ionic bond?
They are shared equally between atoms
They are shared unequally between atoms
They are transferred from one atom to another
They remain with their original atoms
10. Which of the following best describes the structure of ionic compounds?
Molecules with covalent bonds
Lattice structure of alternating positive and negative ions
Random arrangement of atoms
Single atoms bonded together
We recommend that you do not leave the page that you are taking this quiz in. Stay honest 🙂
Can Your Friends Do Better Than You in This Quiz?
Share this quiz with your friends and compare results.
Was this page helpful?
More Popular Chemistry Quizzes:
-
Functional Groups Quiz
-
Basic Chemistry Quiz
-
Thermochemistry Quiz
-
Polar Covalent Bonds Quiz
-
Ionic Compounds Quiz
-
Alkanes Quiz