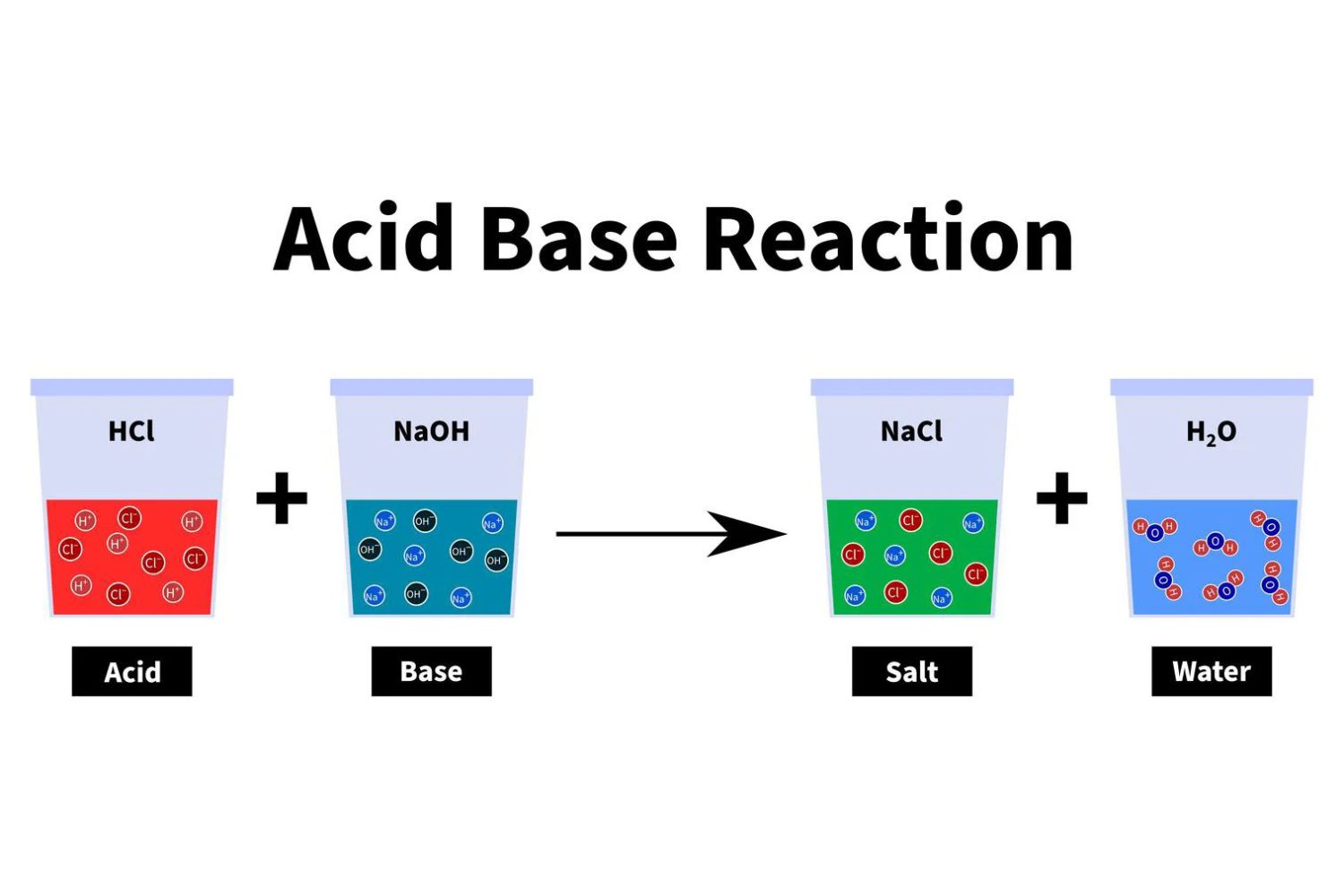
Dive into the fascinating world of acid-base reactions, where molecules interact in ways that are both intriguing and essential to numerous chemical processes. This quiz will challenge your understanding of how acids and bases behave, react, and neutralize each other. Get ready to test your knowledge and deepen your appreciation for these critical concepts in chemistry. Good luck!
We recommend that you do not leave the page that you are taking this quiz in. Stay honest 🙂
Acid-Base Reaction Quiz Questions Overview
1. What is the pH of a neutral solution at 25°C?
0
7
14
1
2. Which of the following is a characteristic of a strong acid?
Partially dissociates in water
Completely dissociates in water
Has a high pH
Is insoluble in water
3. Which of the following is considered a weak base?
Sodium hydroxide
Ammonia
Potassium hydroxide
Calcium hydroxide
4. What is the product of the neutralization reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH)?
Water and sodium chloride
Hydrogen gas and sodium chloride
Water and sodium hydroxide
Hydrogen gas and sodium hydroxide
5. Which of the following indicators turns pink in a basic solution?
Litmus
Phenolphthalein
Methyl orange
Bromothymol blue
6. What is the conjugate base of H2SO4?
HSO4-
SO4^2-
H2SO3
HSO3-
7. Which acid is commonly found in vinegar?
Acetic acid
Citric acid
Lactic acid
Sulfuric acid
8. What is the pH of a 0.01 M solution of HCl?
2
12
4
7
9. Which of the following is a monoprotic acid?
H2SO4
H3PO4
HCl
H2CO3
10. What is the main component of stomach acid?
Sulfuric acid
Hydrochloric acid
Nitric acid
Acetic acid
11. Which of the following bases is used in soap making?
Sodium hydroxide
Ammonium hydroxide
Calcium hydroxide
Potassium hydroxide
12. What is the Bronsted-Lowry definition of a base?
A substance that donates protons
A substance that accepts protons
A substance that donates electrons
A substance that accepts electrons
13. Which of the following is an example of a buffer solution?
A solution of HCl and NaCl
A solution of NH3 and NH4Cl
A solution of NaOH and KOH
A solution of H2SO4 and HCl
We recommend that you do not leave the page that you are taking this quiz in. Stay honest 🙂
Can Your Friends Do Better Than You in This Quiz?
Share this quiz with your friends and compare results.
Was this page helpful?
More Popular Chemistry Quizzes:
-
Functional Groups Quiz
-
Basic Chemistry Quiz
-
Thermochemistry Quiz
-
Ionic Bonding Quiz
-
Stoichiometry Quiz
-
Polar Covalent Bonds Quiz