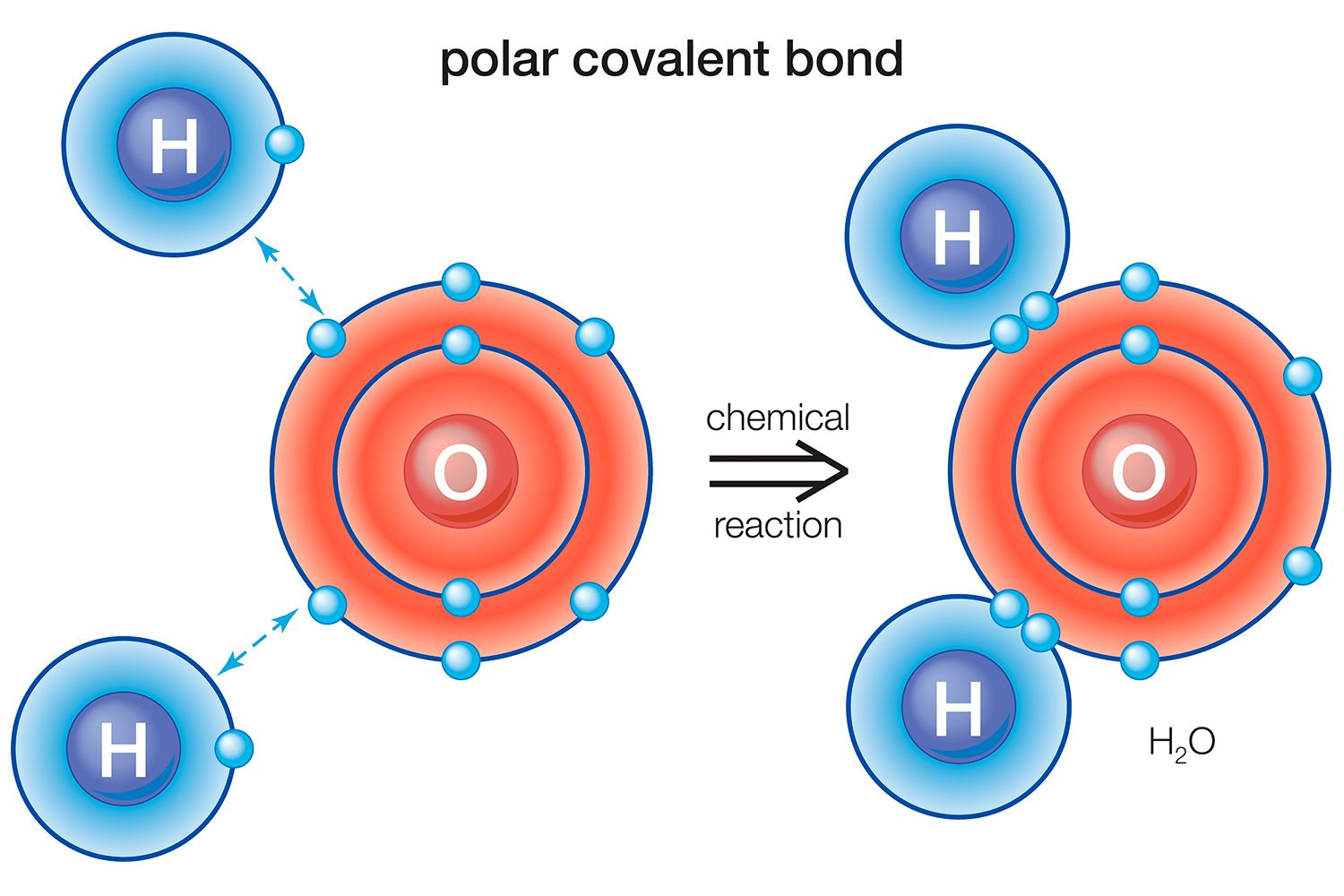
Dive into the intriguing world of polar covalent bonds, where electrons are shared unequally, creating fascinating molecular interactions. This quiz will challenge your understanding of how these bonds form, their characteristics, and their roles in various chemical compounds. Ready to test your knowledge and deepen your understanding of this essential chemical concept? Let’s get started!
We recommend that you do not leave the page that you are taking this quiz in. Stay honest 🙂
Polar Covalent Bonds Quiz Questions Overview
1. What defines a polar covalent bond?
Equal sharing of electrons
Unequal sharing of electrons
Transfer of electrons
No sharing of electrons
2. Which of the following molecules has a polar covalent bond?
H2
O2
HCl
N2
3. What is the result of the unequal sharing of electrons in a polar covalent bond?
Atoms remain neutral
Atoms become ions
Atoms have partial charges
Atoms lose electrons
4. Which property is commonly associated with polar covalent bonds?
High melting points
Conductivity in solid state
Solubility in water
Non-polarity
5. Which of the following pairs of atoms is most likely to form a polar covalent bond?
C and H
Na and Cl
O and H
N and N
6. What term describes the measure of an atom’s ability to attract and hold electrons?
Ionization energy
Electronegativity
Electron affinity
Atomic radius
7. Which molecule is an example of a non-polar covalent bond?
CO2
H2O
CH4
NH3
8. What type of bond is formed between two atoms with identical electronegativities?
Ionic bond
Polar covalent bond
Non-polar covalent bond
Metallic bond
9. Which of the following statements is true about polar covalent bonds?
They always form between metals and non-metals
They result in molecules with no partial charges
They involve unequal sharing of electrons
They are weaker than non-polar covalent bonds
10. What is the primary factor that determines the polarity of a covalent bond?
Atomic mass
Electronegativity difference
Number of protons
Electron configuration
11. Which of the following molecules has a non-polar covalent bond?
O2
H2O
HF
NH3
12. What happens to the electron density in a polar covalent bond?
It is evenly distributed
It is concentrated around the more electronegative atom
It is concentrated around the less electronegative atom
It is lost to the surrounding environment
13. Which of the following best describes the bond in HF (hydrogen fluoride)?
Non-polar covalent
Polar covalent
Ionic
Metallic
14. Which property is NOT typically associated with polar covalent compounds?
High solubility in water
High melting points
Partial charges on atoms
Formation of hydrogen bonds
15. What is the partial charge on hydrogen in a water molecule (H2O)?
Partial positive
Partial negative
No charge
Full positive
16. Which of the following best explains why water is a polar molecule?
Symmetrical shape
Equal sharing of electrons
Bent shape and polar bonds
Non-polar bonds
17. Which type of intermolecular force is strongest in polar covalent compounds?
London dispersion forces
Dipole-dipole interactions
Hydrogen bonding
Van der Waals forces
18. Which of the following elements is most likely to form a polar covalent bond with hydrogen?
Carbon
Oxygen
Sodium
Helium
19. What is the main difference between polar covalent and ionic bonds?
Polar covalent bonds involve equal sharing of electrons, while ionic bonds involve unequal sharing.
Polar covalent bonds involve unequal sharing of electrons, while ionic bonds involve electron transfer.
Polar covalent bonds involve electron transfer, while ionic bonds involve equal sharing.
Polar covalent bonds involve no sharing of electrons, while ionic bonds involve unequal sharing.
20. Which of the following best describes the bond in a molecule of CO2?
Non-polar covalent
Polar covalent
Ionic
Metallic
21. What is the partial charge on oxygen in a water molecule (H2O)?
Partial positive
Partial negative
No charge
Full negative
We recommend that you do not leave the page that you are taking this quiz in. Stay honest 🙂
Can Your Friends Do Better Than You in This Quiz?
Share this quiz with your friends and compare results.
Was this page helpful?
More Popular Chemistry Quizzes:
-
Functional Groups Quiz
-
Basic Chemistry Quiz
-
Thermochemistry Quiz
-
Stoichiometry Quiz
-
Ionic Bonding Quiz
-
Organic Chemistry Quiz